|
Experiential learning resources for the innovative educator
As someone with a background in biology, the first places I would think to go for scientific inquiry experiences is outside or in a lab. Next would be the kitchen. Need some easy kitchen science experiment ideas? Head inside -->
Winter weather in Minnesota can get extreme and those extremes tend to last a while. Cooking is an indoor activity that is loaded with learning opportunities, particularly in science, for those cold or rainy days.
Scientific inquiry is a method of gathering information and gaining scientific knowledge. The students themselves make the observations, ask the questions, make predictions, and experiment. Experimental inquiry is a student-centered way for learners to make discoveries about the world in their own way. The kitchen is a great place to start! This post offers a list of winter-inspired dessert ideas for students to bake while learning the science behind the food through experimental inquiry.
My recommendation is to have students conduct open inquiry. The student directs the inquiry investigation, starting with asking their own questions, They design and conduct an experiment to test one of those questions.
Enjoy the following winter-inspired easy kitchen science experiment ideas! I have done all of these kitchen science experiments with either my own kids (K and 3rd) or my students (8-12 graders). Each is fun, doable, and because they are student-led, modifiable for a variety of ages and skill levels. Easy Kitchen Science Experiment Ideas for All Ages
1. Eggnog
The Question: Does the speed at which eggs are added to hot milk affect the texture of eggnog?
In other words, how do I make chunk-less eggnog? My children and I made homemade eggnog. It wasn't good. My kids despise it in general, but our final result was scrambled, so I certainly wasn't going to convince them to like it that day! The fun in this activity is trial and error. The Science: In order to make smooth eggnog, you have to temper the mixture, which is the process of controlled heating and cooling. Pouring the eggs into the hot milk mixture too quickly is not controlled. Heat coagulates the proteins in the eggs, so adding the eggs to the heat too quickly causes them to scramble. Tempering, slowly controlling temperature, keeps the protein structure intact. Inquiry Activity: Ask students to explore the ingredients and the process of making eggnog. They can make observations, ask questions, and design their own experiments. Click here for the recipe that we used. Or they can experiment with the question posed here (Does the speed at which eggs are added to hot milk affect the texture of eggnog?). They can try it out and experiment with different methods.
2. Tye-Dye Sponge Roll Cake
The Question: Which ingredients prevent a "roll cake" from crumbling when rolled?
This was such a fun recipe to make with kids. My kiddos made two different types of cake to see how particular ingredients (or lack thereof) might impact the cake texture, making it more or less suitable for rolling without cracking. We made a roll cake and a regular white cake. The Science: A mixture of eggs and sugar provides structure, in part by trapping air bubbles during the whipping process. In addition, adding baking powder causes a chemical reaction resulting in carbon dioxide, increasing the number of bubbles in the batter. Proteins and starch in the flour add stability to the air bubbles, but the high ratio of sugar to flour gives it that flexibility that roll cakes require. Inquiry Activity: Ask students to explore the ingredients and the process of making roll cakes. They can make observations, ask questions, and design their own experiments. They can make a variety of different types of cakes, experiment with baking powder, make a control roll cake and a roll cake missing one ingredient, etc. I used this recipe for the roll cake.
3. Pie Crust
The Question: Which fat makes a flakier pie crust, butter or lard?
My high school advisory students started an annual holiday pie fundraiser many years ago, and part of the planning process was creating the BEST pie to sell. We asked around, talked with professional chefs, surveyed consumers, perused caking blogs, and conducted experiments. The ultimate influence was my grandmother! She said the secret to her flaky pie crust is animal lard. So we tested it. The Science: Lard is fat. Period. Butter, on the other hand, is fat plus some water. Lard has a higher melting point than butter, making it easier to handle in the making process. Lard also results in a flakier crust because the 100% fat in the lard solidifies into crystals, separating the dough into layers, and creating that flakiness we were testing for. Inquiry Activity: Whether flakiness is desired is a matter of opinion. But we did find that lard, in fact, does make pie crust flakier, whether we prefer that texture or not. Learners can investigate their own questions about variables that inspire or interest them. They might investigate why pie crust recipes usually ask for cold water versus hot or room temperature water. Students might experiment with different types of flour such as whole wheat, all-purpose, and pastry flour. Students could play around with crust width, baking temperature, fillings, glazes, vents created for baking, and so on and so on. Find any number of pie crust recipes on Google. Note: Lard is not required to make pie crust! Learn about your students and be sensitive to their needs. Allergies, food sensitivities, religious food practices, etc. need to be considered and respected.
4. Whipped Cream
The Question: Which milk product will result in the foamiest texture for whipped cream - skim milk, whole milk, or heavy whipping cream?
This question came up because we wanted to make whipped cream for the pie that we made, but didn’t have heavy whipping cream on hand. We had whole milk. A student wondered if it really mattered. So again, we tested it. The Science: The heavy whipping cream turned out to be incredibly important. No matter how long we blended the milk mixtures, they never became frothy. The fat in the heavy whipping cream envelopes air bubbles added from the whipping process, creating a network that stiffens the mixture. So, fat is what gives it the foamy texture, and whole milk just wasn’t fatty enough. Inquiry Activity: Ask students to explore the ingredients and the process of making whipped cream. They can make observations, ask their own questions, and design their own experiments. This is experimental inquiry, experiments driven by students’ observations and questions. Or learners can design an experiment that tests the question I asked above. Click here for the recipe that we used.
5. Cream Puffs
The Question: Does the number of eggs affect the structure of cream puffs?
My kids and I have made many bread-type recipes, but this one called for an unusual number of eggs. We made three batches of cream puffs, some with the number of eggs suggested in the recipe, one with half of the number of eggs suggested in the recipe, and one with no eggs at all. The batch without eggs collapsed completely. The Science: Cream puffs also have a lot of fat, so egg protein is necessary to keep the structure. Eggs also act as a leavening agent and an emulsifier for a smooth and light final texture. Inquiry Activity: Ask students to explore the ingredients and the process of making cream puffs. There is so much science involved in these little desserts! For this reason, I would encourage open inquiry. Otherwise, you can offer up a specific question, such as the one above. I found there to be a few unusual cooking methods in this recipe, such as melting the butter in boiling water and milk first. Those types of investigations are fun as well. Try this recipe. Combine this cooking activity with the whipped cream inquiry above for a really tasty treat!
6. Hard Candy
The Question: Does the temperature of the sugar mixture when removed from the heat source impact the hardness of the final product?
This is a question that my high school students and I investigate every year. I am hesitant to make hard candy with my own children, who are 5 and 8, because of the burning risk. I burned myself making this candy three days before my wedding! This recipe is probably more appropriate for older learners, but regardless of age, safety should be a priority and taken seriously. The Science: Hard candy ingredients include only sugar and water (and flavor extracts and food coloring if you wish). You combine the sugars and water and boil the mixture until it reaches 300° F (hard crack). Boiling the mixture to this specific temperature results in the evaporation of most of the liquid, leaving behind a lot of sugar, thus it sets as hard candy. If the mixture only reaches 250° F, in contrast, not enough water will have evaporated to harden the sugar, leaving a softer consistency such as that of toffee (hard ball), or 270° yielding a taffy-like consistency (soft crack). If the mixture over-evaporates, reaching 320° F, for example, the liquid sugar will caramelize. Temp is critical, and evaporation is the simple scientific explanation for that. Inquiry Activity: Ask students to explore different evaporation times (cooking temperatures) to test the question that I mentioned above. They could also investigate their own questions and experiment with variables other than temperatures such as ingredient quantities or types of sugar (raw sugar vs. white, glucose syrup vs. corn syrup, etc.) Click here for the recipe that we use. I always add flavor extracts to get into the winter spirit, such as peppermint and cinnamon.
7. Gingerbread Cookies
The Question: Does the amount of time that cookie dough chills in the refrigerator impact the shape and texture of the cookies?
This was a question asked by my kindergartener, only it was more like, "why do we have to put this cookie dough in the refrigerator, I want to make them now!" We were able to explore this question together with my assistance. We simply made two batches of cookies, one with dough that we didn't chill and one with dough that was refrigerated for two hours. It would have been better if we added a couple more batches, one chilled for 20 minutes and one for 12 hours, for example. The Science: Turns out that the fats in the butter solidify when chilled, giving it more structure. This was important to keep its shape during the rolling, cookie-cutting, and baking processes. You can see an obvious difference in the photos between the cookies that were chilled and those that were not. The non-chilled cookie dough cookies were essentially shapeless blobs. It also takes longer for the butter fat to melt in the chilled cookie during the baking process, making the final product slightly chewier and softer. Inquiry Activity: Ask students to explore the ingredients and the cooking process of gingerbread cookies. Sugar cookies have the same chill requirement, so you can simplify it a bit by skipping the ginger and molasses required in gingerbread. Learners can make observations, ask questions, and design their own experiments to test the questions. Click here for the recipe that we used.
8. Marshmallows
The Question: What gives marshmallows a smooth, foamy, stretchy consistency?
Making marshmallows came up once by accident while my students and I were making hard candy (mentioned above). Our candy thermometer was broken. So we tried to guess. We significantly underestimated the temperature of the sugar/water mixture when removed from the heat source, which resulted in a squishy, stretchy substance comparable to marshmallows. After some investigating, however, students discovered that marshmallows have a key ingredient, however, that other types of sugary candy-like sweets lack. The Science: That ingredient is gelatin. Water, sugar, and corn syrup are heated on the stove. Some of the liquid evaporates. The remaining sugar/water combo is removed from heat, slowly added to a gelatin/water mixture, and beaten, which adds air bubbles to the mixture. As this blended foam cools, the corn syrup prevents crystallization and the gelatin turns from a liquid to a gel, trapping bubbles inside. This special and specific combo of ingredients and cooking method gives marshmallows their structure and consistency. Inquiry Activity: Students can ask their own questions about marshmallows and conduct open inquiry experiments, or they could explore something specific such as the importance of temperature, sugar quantity, gelatin quantity, the timing of adding gelatin in the making process, the type of sugar used, and more. Learners can even experiment with marshmallows that they have already made, such as the hot chocolate temperature required to melt a marshmallow, or how the amount of time in a microwave affects the shape and consistency of a marshmallow Click here for the recipe that we used. There are many others!
9. Meringue
The Question: Does the blending speed impact the stiffness of meringue peaks?
My students and I discovered the answer to this question by pure accident. My high school advisory students participated in our annual school pie-baking contest. They decided on lemon meringue pie, but we didn't have a blender on hand. We hand-whisked the egg-white mixture. It took a very, very, very long time, but we did eventually get those peaks. But I wouldn't have called them stiff. I have made many meringues since but used a blender. I highly recommend it. The Science: As you blend egg whites, air bubbles form. The proteins from the egg whites break up from the blending and rearrange themselves, surrounding the air bubbles and securing them, leaving a foamy consistency. By blending too slowly or weakly, the proteins will not "denature", and thus will not incorporate the scaffolding necessary to keep air bubbles in place. Or by whisking limply, you may not be adding any air bubbles to the mixture at all! You can absolutely hand-whisk very quickly, but expect a labor of love! Inquiry Activity: Ask students to explore the ingredients and the cooking process of meringue (cookies, pies, etc.) They can make observations, ask questions, and design their own experiments, such as why sugar is added slowly, the result of over-whipping, using whole eggs instead of egg whites, leaving out the cream of tartar, the ideal baking temperature, etc. Click here for the recipe that we used.
10. Chocolate Dessert Bowl
The Question: Does the quality of the chocolate affect the texture after it has been tempered and hardened?
My own children and I tried to make a chocolate bowl using a Pinterest recipe, and it didn't work out. AT ALL. We wondered where we went wrong, and if the type or quality of chocolate mattered. The Science: There is cocoa butter in chocolate, which is fat. The fat forms crystals when the chocolate is tempered (controlled heating and cooling of the chocolate). Tempering chocolate breaks up crystals made from the fat and creates a new, more orderly crystalline structure, giving it a glossy look in the end. We wondered if a higher quality chocolate, one with better or different amounts of cocoa butter would make a difference. Inquiry Activity: We never tested our question, but think it would be a great way to start kitchen science experimentation, especially for beginners. For those that have more experience with inquiry science, encourage open inquiry investigations. Ask students to explore chocolate in general, not necessarily a specific recipe. What gives it its shape? What is the difference between chocolate syrup and the chocolate on a candy bar? How is chocolate made? Etc.
I don't know about your students, but most of mine love to cook or bake. Even those that don't really like to cook can find entertainment from these easy kitchen science experiment ideas.
I highly encourage that you don't plan their experiments for them. You may have noticed that most of the experiment ideas listed in this post came from observations, obstacles, or student questions. Many of them came about by pure chance. If you can, give your students the freedom to explore and develop their own kitchen science experiments with you as a guide. Good luck, and happy baking!
More Science Blog Posts:
Experimental Inquiry Resources:
Join our experiential learning Facebook group!
Did you know there is an experiential learning Facebook group? Check that out - Experiential Learning Community for K12 Teachers - and join in the discussion about experiential learning ideas! Find us on social media! Follow Experiential Learning Depot on Pinterest, Facebook, Youtube, and Instagram for more on experiential education, and check out my shop for experiential learning resources. Observe. Question. Explore. Share.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
Blog IntentTo provide innovative educational resources for educators, parents, and students, that go beyond lecture and worksheets. AuthorSara Segar, experiential life-science educator and advisor, curriculum writer, and mother of two. Categories
All
|






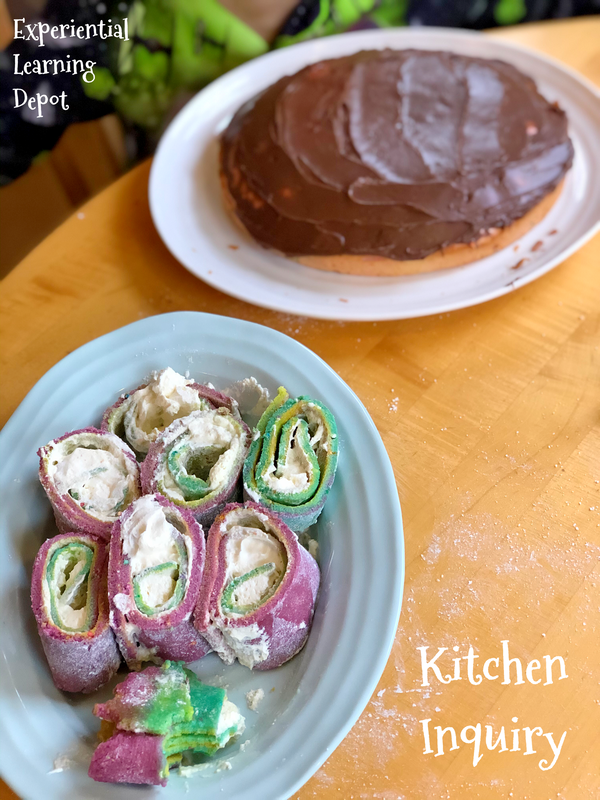







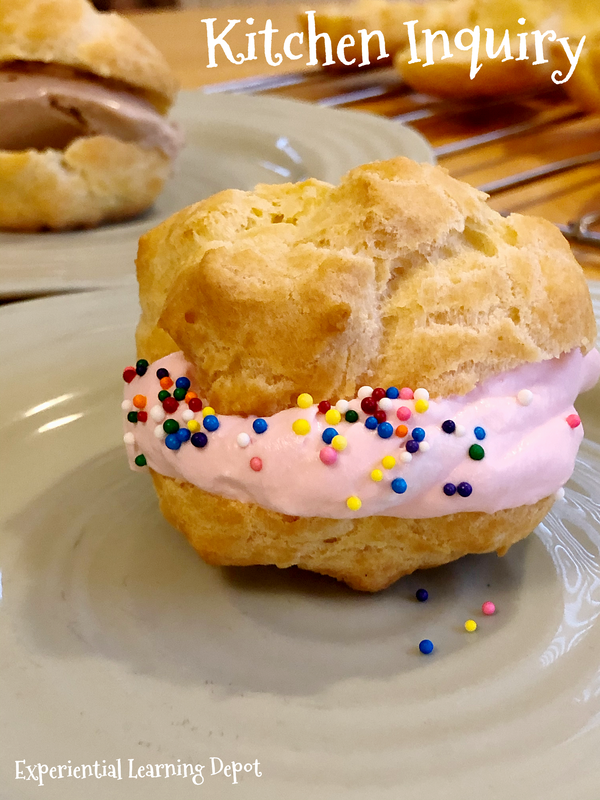

























 RSS Feed
RSS Feed